Scientists at the John Innes Centre have discovered how plants make valuable natural products we rely on today for use as medicines, flavours and scents. This discovery has the potential to pave the way for the creation of entirely new drugs, flavourings and cosmetic ingredients.
Most plant-derived drugs, scents and flavours contain hydrocarbon rings in their structures – and until now exactly how Nature makes them has remained a mystery.
Recent work by a team of scientists at the John Innes Centre, led by Dr Paul O’Maille, resulted in the discovery of the origins of these cyclic or ring forming reactions in plants – which yield medicines like artemisinin: the most potent antimalarial drug, as well as flavours such as ginger and scents like bergamot.
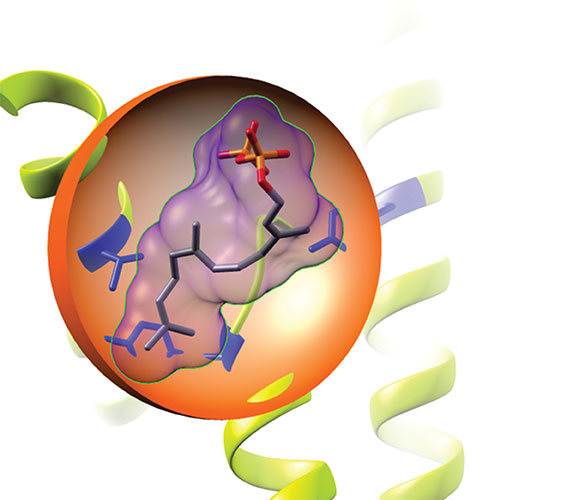
The key to their success was breeding enzymes, the protein machinery that catalyses chemical reactions in plants. In particular they focused on enzymes that catalysed the formation of terpenes: the most diverse class of predominantly cyclic (ring-containing) natural products. By breeding a pair of enzymes, one that makes linear, less complex terpenes with one that makes cyclic terpenes, Dr Melissa Salmon, the lead author on the paper, was able to localize the trait of cyclization in the protein structure.
In their landmark discovery, the team of scientists identified a natural ‘gateway’ mutation that unlocks cyclization. Furthermore, they went on to identify a network of additional natural mutations that work together to turn cyclization on and off. Using this network of mutations, they were able to activate cyclization in other linear terpene producing enzymes, hinting that nature may rely on these synergies to make new enzymes and hence new natural products.
“It is fascinating to see that nature has placed fine control of this key process in the hands of a delicate network of interactions, a mechanism that has parallels with the internal mechanism of a lock; by manipulating this we can now unlock chemical diversity” said Dr Jitender Cheema, a computational biologist on the team.
Dr O’Maille said the discovery was a pivotal moment in his lab: “We saw a real explosion of chemical diversity coming out of our enzyme breeding experiments. For a scientist like me, studying the origins of chemical diversity, this is like discovering signatures of the Big Bang.”
Understanding how cyclization pathways emerge in nature will pave the way for developing new drugs, flavours and fragrances. Also by unlocking the molecular mechanism by which these rings form, we could increase efficient production of high value compounds already in use by pharmaceutical, cosmetic and food industries.
This research was funded by the Biotechnology and Biological Sciences Research Council (BBSRC) and published in Nature Communications.
Dr O’Maille’s team has begun testing activity of some terpene synthase-controlled cyclic products against both human pathogens like Helicobacter – the bacterium pathogen responsible for stomach ulcers and plant pathogens like Phytophthora infestans which causes the potato blight.
Source
John Innes Centre, press release, 2015-02-03.
Supplier
Biotechnology and Biological Sciences Research Council (BBSRC)
John Innes Centre
nature (Journal)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










