 Without the help of enzymes, we couldn’t digest food or synthesize DNA: They make the right metabolic reactions happen at the right speed.
Without the help of enzymes, we couldn’t digest food or synthesize DNA: They make the right metabolic reactions happen at the right speed.
But like Goldilocks, enzymes won’t tolerate conditions that are too hot or too cold. They typically function within a narrow temperature band.
To give them greater range, and potentially more applications, a team of Yale engineers led by assistant professor of chemical and environmental engineering Corey Wilson has developed a method for designing temperature adaptive enzymes.
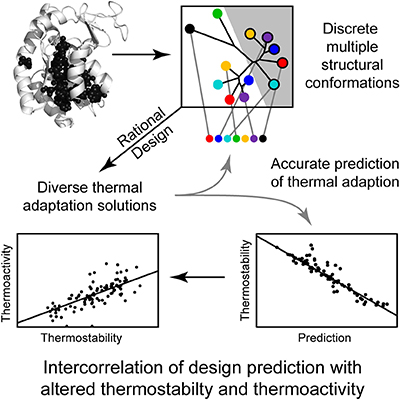 The innovation, believed to be the first of its kind, complements previous experiments focused solely on enzyme structure and flexibility, the sequence of the enzyme’s amino acid building blocks, or how the enzyme utilizes energy.
The innovation, believed to be the first of its kind, complements previous experiments focused solely on enzyme structure and flexibility, the sequence of the enzyme’s amino acid building blocks, or how the enzyme utilizes energy.
Using Wilson’s method, enzymes can be designed to function at specific temperature ranges.
“We can now make enzymes that can be used at higher or lower temperatures and under harsh conditions,” says Wilson. “You might say to me, ‘Look, I need to catalyze this industrial reaction at a hundred degrees, I need a system to do this catalysis.’ Now we can create an enzyme to do that.”
For example, an enzyme could be modified for use in biofuel production, where the high temperature (approaching 200 degrees Celsius) pretreatments would cause the currently available enzymes to lose their structure.
Alternatively, for patients whose body temperatures have been artificially lowered during cardiopulmonary bypass surgery, an enzyme could be designed to more efficiently return thinned blood to its normal state, thereby improving safety.
In addition to such applications, temperature adaptive enzyme design has also enabled the team to accurately identify the thermodynamic limits of protein function. Given merely a fingerprint of a protein or the hint of a protein, the new method could theoretically predict the environments in which an organism might live.
“You can extrapolate that to anyplace,” Wilson says. “You can go off the planet into the universe and you can say here are the conditions that would support this kind of enzyme. It can be used as a predictive tool to determine the thermodynamic limits of life.”
Other members of Wilson’s team include Stanley C. Howell, Krishna Kishore Inampudi, and Doyle P. Bean, all of Yale. Their research was recently published in the journal Structure.
Source
School of Engineering & Applied Science (Yale University), press release, 2014-01-13.
Supplier
School of Engineering & Applied Science | Yale University
Yale University
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










