As carbon dioxide levels in the Earth’s atmosphere increase, average global temperatures climb. 2015 was the hottest year on record, with 2014 a close second. These warm temperatures bring with them drastic effects on our climate. (For more information on climate change, check out our special edition on climate change from 2014). These giant, Earth-sized problems could be mitigated by some of the smallest living beings: microorganisms. New research from the Stephanopoulos group at MIT reports engineered microorganisms that can generate sustainable biofuel from carbon dioxide emissions.
Where do carbon emissions come from, and what do we usually do with them?
First, let’s understand the source of the carbon dioxide emissions. These emissions come from the combustion of carbon-based “fossil” fuels, like coal, oil, and natural gas, to produce energy. Unfortunately, carbon-based fuels are so integral to our global energy system that “decarbonizing” our energy use is a monumental task. Two-thirds of power plants worldwide would have to be replaced with renewable electricity sources, like wind turbines and solar panels. We would need alternative ways to make steel, aluminum, and concrete, as the production processes of each of these materials emits carbon dioxide. Oh, and each of the billion or so cars on the road would have to be traded in for an electric model. And every gas stove or heater would have to be electrified as well. Even if we achieved all of this, we’d soon run into challenges like air travel that can’t be electrified without dramatic technological innovations
Scientists have started looking for ways to capture carbon dioxide before it reaches the atmosphere and store the gas deep underground, where it can’t do any harm. But attaching carbon capture and storage (CCS) capabilities to a standard coal or natural gas power plant adds more than 25% to the cost of electricity from the plant Essentially, electricity producers lack economic incentives to reduce carbon dioxide emissions via CCS. The Holy Grail here would be some technology that takes in carbon dioxide and converts it into a marketable product.
Using microorganisms to produce liquid fuel
One technology in particular, being developed by Greg Stephanopoulos’s group at MIT, is a method for turning industrial waste gases like carbon dioxide into fuel. Their research was recently published in the Proceedings of the National Academy of Sciences. This conversion process starts with mixtures of carbon dioxide and either carbon monoxide or hydrogen, The gas is bubbled into a two-step reactor (Figure 1), in which carefully chosen microorganisms digest the waste gases to produce lipids, or fats, which are easily converted into a diesel fuel substitute called biodiesel. The process consumes carbon dioxide, preventing it from reaching the atmosphere, and it uses cheap (or even free) ingredients to produce a marketable product.
Let’s look a bit deeper into how this works. In the first stage, the gaseous mixture is fermented by Moorella thermoacetica, a species of bacteria that can be found naturally in ponds and undersea vents. The M. thermoacetica consumes carbon dioxide and hydrogen for energy, forming acetic acid, or vinegar, as a waste product. The acetic acid is then pumped into the second stage, where a second microorganism, the yeast Yarrowia lipolytica, munches on the vinegar. The yeast has been engineered by the MIT team to produce lots of lipids, or fats, from the acetic acid. These lipids can easily be converted into biodiesel by mixing them with an alcohol, like methanol or ethanol, in the presence of a base such as sodium hydroxide (an active ingredient in Drano).
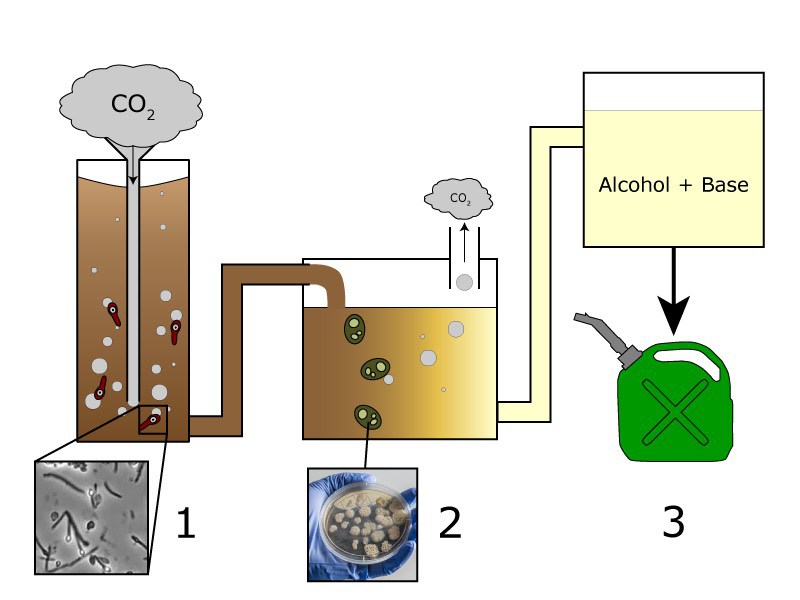
According to the MIT team, the first stage in the process should consume twice as much carbon dioxide as the second stages releases. This calculation doesn’t take into account the carbon dioxide produced when the fuel is burned, but it does represent a step towards decreasing our overall carbon footprint, because the procedure is a net consumer of carbon dioxide.
This new process has advantages over other biofuel production methods in the availability and cost of their feedstock, or chemical ingredients. Usually, the feedstocks are the most expensive part of producing biodiesel. The high cost arises because most biodiesel feedstocks are either sugar cane or corn, which are in high demand as food items. Ramping up biodiesel production could increase demand for these foods and further raise their prices, potentially making food too expensive for low-income families. By designing a process which uses waste gases, the MIT team has found a way to use inexpensive feedstocks to make a valuable product and sidestepped the issue of increasing food prices.
What’s the impact of this technology?
So, is this technology going to save our planet? First, the MIT team will have to prove the reaction works at scale. They have plans for a 20,000 L reactor in the works, capable of producing 30-40 gallons of biodiesel per day.
But how far would 30-40 gallons of biodiesel move a car? A gallon of biodiesel and a gallon of gasoline have roughly the same energy content, so you can think of them similarly. The 20,000 L plant, then, could refuel 2-3 cars a day. But this is a drop in the fuel tank compared to gasoline usage. Americans consume a hundred gallons of gasoline for every gallon of biodiesel (figure 2), which adds up to almost 400 million gallons of gasoline per day. Assuming a 16 gallon average tank size, that’s 25 million cars that need refueling every day, so we’d need ten million of these 20,000 L biodiesel plants.
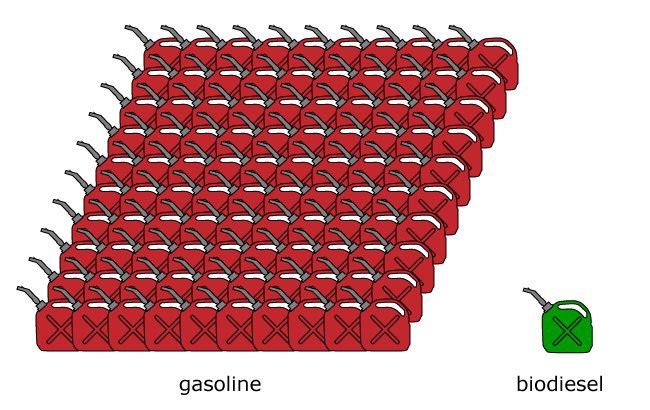
To put ten million biodiesel plants in perspective, consider that there are only about 125,000 gas stations in the United States – so we’re talking about 100 biodiesel plants for every gas station. We’ll need more ways of cost-effective, sustainable fuel production to meet this demand. A host of technologies, in various states of research and development, could potentially pitch in. For example, a biotech startup, is working towards producing sustainable fuel, also with the aid of microorganisms. Joule’s pilot plant in New Mexico (video) uses bacteria capable of photosynthesis to produce ethanol from carbon dioxide. They hope to scale up their existing plant to 25 million gallons per year, and have identified potential sites for new plants.
The combination of carbon capture and storage with renewable fuel production could revolutionize our world. New jobs would be created as the industries expand. Nations could ensure energy independence and security by producing their own fuels. We could slow or even halt the march of global warming, reducing the threat of sea level rise.
And we could make some microscopic friends while we’re at it.
Michael R. Gerhardt is a Ph.D. candidate in Applied Physics at the Harvard John A. Paulson School of Engineering and Applied Sciences.
Author
Michael R. Gerhardt
Source
Harvard University, press release, 2016-05-26.
Supplier
Harvard University
Joule Unlimited, Inc.
Massachusetts Institute of Technology (MIT)
Proceedings of the National Academy of Sciences of the USA (PNAS)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










