At NC Partnering we hold the confident view that wood and other prime biomass should be utilised for maximum value creation in the bioeconomy. In past Newsletters and speeches, we have argued against playing the chemicals game on the petrochemical industry’s terms, through so-called drop-in solutions where an established petro-product is replaced in isolation by a functionally similar bio-product. Our arguments have been based on common sense and economic considerations. We are therefore delighted to now bring a leading chemistry scientist’s take on this vital subject.
 Dr Thomas Farmer is doing groundbreaking work at the University of York in bio-chemicals, very much laying the foundation for tomorrow’s best practices. I this first of two articles, he outlines how bio-chemicals need to be derived more through reduction than oxidation, the latter being a sort of default setting as it is the norm in most petrochemical processes.
Dr Thomas Farmer is doing groundbreaking work at the University of York in bio-chemicals, very much laying the foundation for tomorrow’s best practices. I this first of two articles, he outlines how bio-chemicals need to be derived more through reduction than oxidation, the latter being a sort of default setting as it is the norm in most petrochemical processes.
NC: Does the fact that ‘only’ about 10% of total raw oil is converted into petrochemicals indicate that there is serious opportunity for bio-based chemicals to become equally or even more used in the foreseeable future?
TF: I think it most certainly does. Although the chemical industry only takes <10% (by volume) of the total annual production of crude oil it goes on to produce nearly half of the wealth from petroleum [1,2]. As such this shows that there is greater value in producing chemicals than fuels or energy from a resource, and the same will most likely apply when using biomass. By converting biomass to chemicals rather than fuel and energy we will be able to create more wealth, more jobs and greater prosperity
Of course mankind will always need fuel and energy, and likely always in greater volumes than we need chemicals, but certainly our burgeoning bio-based community should focus primarily in chemical production. It is true that annually the Earth produces vast quantities of new biomass, but we must always remember that most of this cannot easily be diverted to fuel, chemical and energy production. The bio-based chemical industry therefore represents an excellent opportunity to produce value from the “available” biomass, ideally using only waste biomass! It is however on this consideration that perhaps the views of various nations in Europe will differ greatly. There is no doubt that for the UK we must focus our efforts of using waste biomass as our resource for a more sustainable chemical industry as we simply do not have the available free land to divert to biomass production solely as a feedstock for chemicals. In Scandinavia the situation will be very different, where vast areas of land can, and in some instances already are, diverted entirely to the production of say wood for use in material and chemical production, such as that demonstrated by the pulp and paper mills.
There is also a functionality aspect to consider for the fuel vs. chemicals debate. By this I refer to the functional groups of the chemicals we can produce versus the functional groups in feedstocks. The typical molecular structure of base chemicals (i.e. the fundamental building-blocks of the petrochemical industry such as ethene and xylene) are devoid of any heteroatoms (oxygen, nitrogen, chlorine etc.). The basis of the current petrochemical industry is to add oxygen, nitrogen, chlorine and sulfur to these base chemicals, eventually resulting in highly functionalised products used across a vast spectrum of applications. The scenario for bio-based products in very different, and instead we will look to remove oxygen atoms from the molecular structure of the feedstock (such as cellulose, starch and glucose). This concept can be represented graphically via Van Krevelen diagrams like those below (Figures 1-3), where a comparison of petroleum base chemicals vs. bio-derived building blocks (so-called platform molecules) are clearly lcated on different areas of the map.
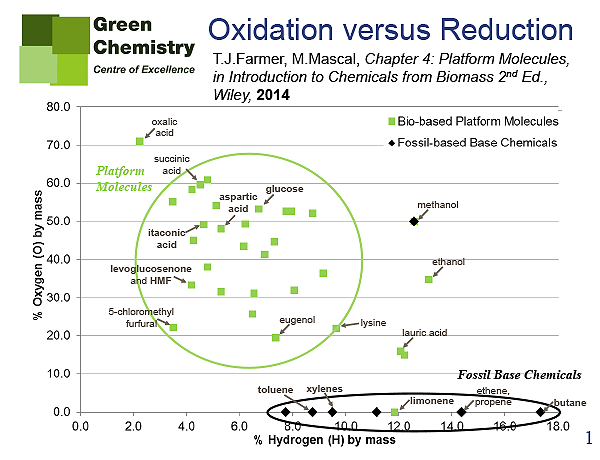
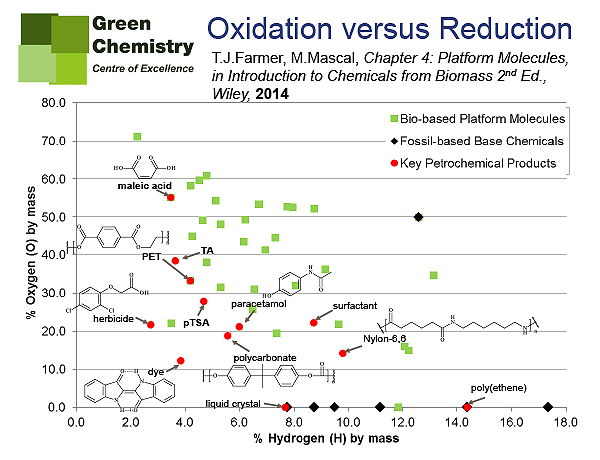
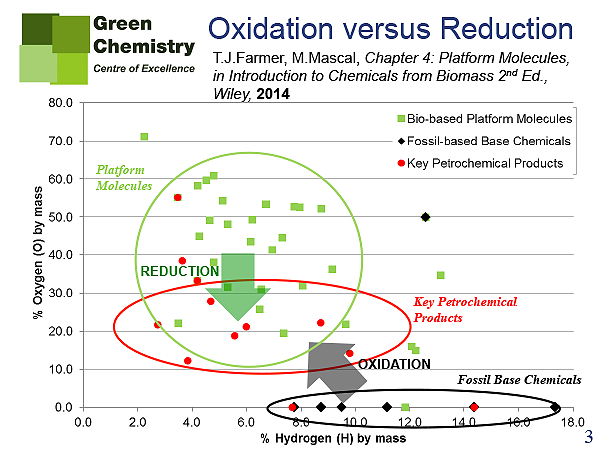
NC: Your findings seem to be pushing the search for economically viable base bio-chemicals away from oxidation towards reduction driven processes. How economically competitive do you envisage these methods becoming in the next decade, and can you indicate which of these products will reach wide commercialisation and global market share?
TF: I certainly feel very strongly that the bio-based chemical industry must seek to use the inherent functionality of biomass better, and this primarily refers to keeping as much of the oxygen content from the feedstock in the final product. If you take the example of converting glucose (C6H12O6) to ethene, via ethanol, you would have to remove all oxygen originally present in the starting material, and this represent a significant proportion of the starting mass of your feedstock being under-utilised. Our Van Krevelen like diagram (Figure 4) clearly highlights this and would suggest that this is not a wise use of the biomass feedstock.
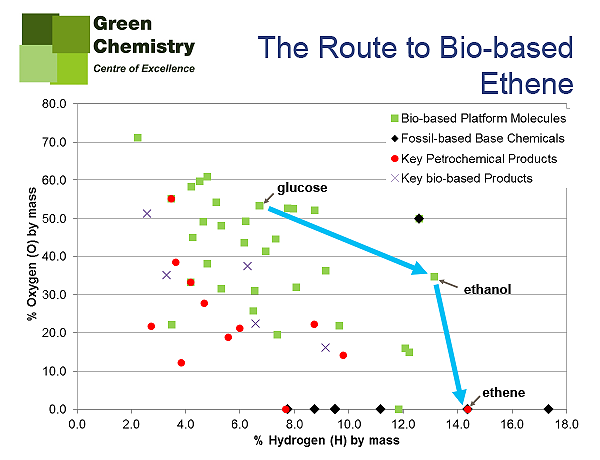
Our current petrochemical industry has had nearly a century to optimise most of the processes it uses to add atoms such as oxygen, nitrogen and chlorine to the base chemicals. Therefore it would be unfair on the researchers working towards to removal of these elements from biomass feedstocks to be able to compete with the petrochemical industry in such as short space of time.
Source
NC Partnering Newsletter, 2016-08.
Supplier
NC Partnering Ltd
University of York (UK)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










