A research group, consisting of doctoral student FUJIWARA Ryosuke, Associate Professor TANAKA Tsutomu (both of Kobe University’s Graduate School of Engineering) and Research Scientist NODA Shuhei (RIKEN Center for Sustainable Resource Science), has succeeded in improving the yield of target chemical production from biomass. They achieved this through metabolically engineering the bacteria used in bioproduction, so that it would use different kinds of sugar absorbed from the biomass for separate aims.
There are problems encountered when using microbes to produce target chemicals; if the microbes use the carbon sources (sugars) for their own propagation, target chemical production decreases. On the other hand, suppressing this propagation causes the microbes to weaken, resulting in an overall decline in production. To try to solve this issue, the research team developed a new strategy called Parallel Metabolic Pathway Engineering (PMPE), allowing them to control both target chemical production and microbe propagation. They used this approach to alter E. coli bacteria in order to successfully boost the production of the nylon precursor muconic acid.
If it becomes possible to utilize the selected carbon source solely for target chemical production and use the remaining sources for microbe propagation, this will bring about great advances in the production of aromatic compounds and raw materials for medical and chemical products.
The results of this research were first published in ‘Nature Communications’ on January 14.
Main points
Development of the PMPE strategy which allows the utilization of sugars for microbe propagation and target chemical production to be independently controlled. Using this approach, the research group successfully increased the yield of muconic acid (the target chemical).
PMPE can be applied to the production of various raw materials, such as aromatic compounds and dicarboxylic acid, utilized in chemical products and medicines.
Expected to improve the effective utilization of raw materials, such as biomass, which contain multiple sugars.
Research Background
We rely on fossil fuels as raw materials for producing various products. However, producing petroleum-derived compounds increases the amount of atmospheric CO2, causing a multitude of environmental problems such as global warming.
Consequently, there is a need to develop biorefinery technologies (*1), which involve using microbes to produce chemical compounds from naturally abundant renewable resources such as tree and plant matter. Biomass-derived products have the advantage of being carbon neutral (*2); they do not increase the amount of CO2 in the atmosphere. It is hoped that using biomass to produce various useful compounds can form a basis for a low carbon society, reducing the amount of atmospheric CO2.
Muconic acid is a useful chemical that can be easily converted into adipic acid, an ingredient in nylon production. It is also used as a raw material in the production of various medical and chemical products. However, it is currently chemically synthesized from petroleum resources. It is hoped that a fermentation method could be developed using microbes and renewable plant-based resources with milder reaction conditions and fewer by-products.
However, there are problems with using microbes to produce target chemicals from biomass. There are many cases where even though the microbes utilize the biomass, they propagate themselves instead of producing the target chemical. However, altering the metabolism to prevent the microbes from increasing causes them to weaken, meaning that the target chemicals cannot be synthesized. The balance between microbes’ self-propagation and target chemical production is big issue.
To solve this dilemma, the research team developed a new technique called Parallel Metabolic Pathway Engineering (PMPE) in which they separated sugar utilization between microbe propagation and target chemical production, allowing them to control each process independently.
Research Content
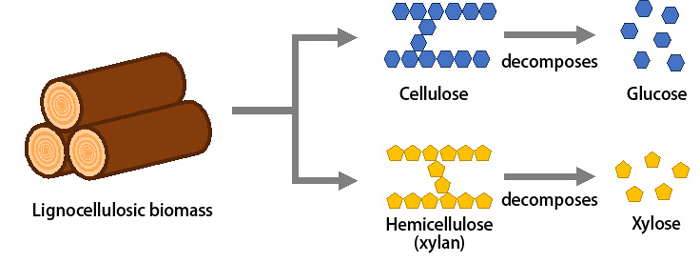
Lignocellulosic biomass, which does not compete with global food supplies, is made up of glucose and xylose sugars (Figure 1). The research team developed a metabolic strategy which involved modifying the E. coli bacteria so that it would utilize glucose for target chemical production and xylose for microbe propagation.
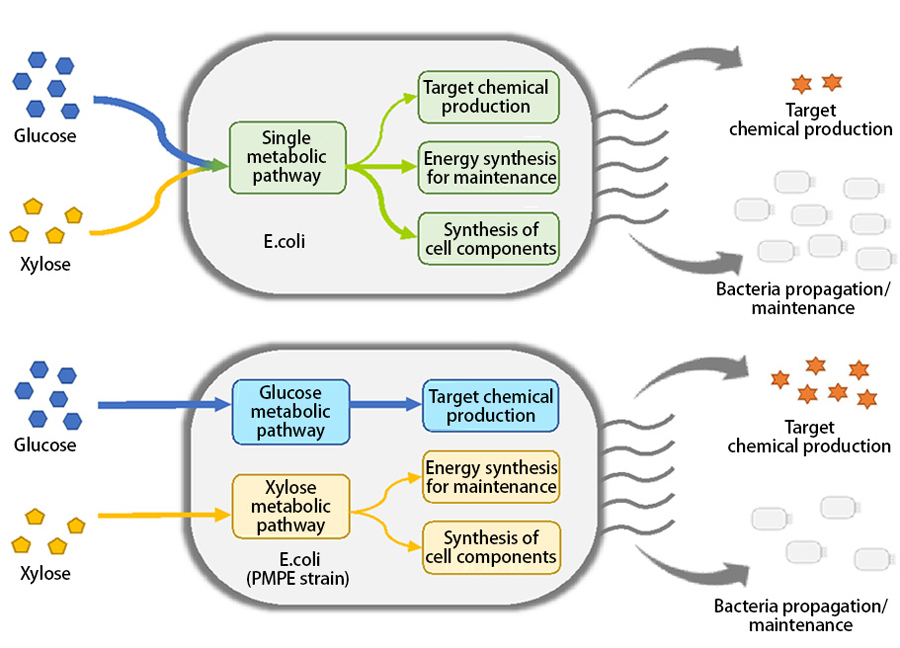
In regular microbes, glucose and xylose use the same metabolic pathway and are both utilized for microbe growth and target chemical production (as shown in Figure 2). This reduces the amount of target chemical synthesized because the microbes absorb the sugars to produce and maintain the elements and energy that they require to live.
To mitigate this issue, the research group developed a new strategy called PMPE. As shown in Figure 2, dividing the microbes’ metabolic pathway allows each sugar to be utilized independently with all the glucose being used for target chemical production and all the xylose being used for microbe propagation and maintenance. This allowed a greater yield of the target chemical to be produced because none of the glucose was being utilized for microbe growth.
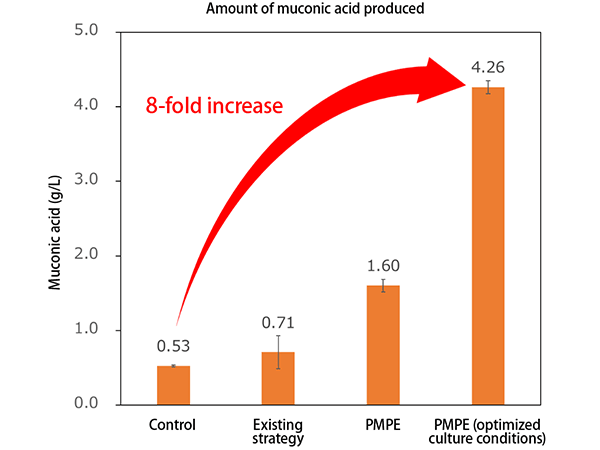
This research group introduced a metabolic pathway to the modified E. coli for synthesizing muconic acid. The modified E. coli utilized the glucose and xylose, leading to the production of the target chemical. The researchers succeeded in producing 4.26 g/L of muconic acid with a yield of 0.31g/g-glucose (Figure 3). This is considered the highest yield in the world, proving the effectiveness of the PMPE strategy.
Subsequently, the researchers investigated whether the PMPE strategy could be applied to the production of target chemicals other than muconic acid. As a result they successfully increased yields of the essential amino acid and aromatic compound phenylalanine, and 1,2- propanediol, which is used as an additive in medicines and food products. These results have shown that PMPE is a versatile technique that can be used to efficiently produce a variety of compounds.
Further Developments
It is expected that the PMPE technique developed by this research group can be applied to increase the production of wide variety of raw materials, such as aromatic compounds and dicarboxylic acid, used in medical and chemical products. Furthermore, this strategy of altering the bacteria’s metabolism will allow biomass containing multiple sugars to be more efficiently utilized.
Acknowledgements
This research received support from the following organizations:
The Japan Science and Technology Agency (JST)’s Mirai program: Small start-type (feasibility study) “Realization of a Low Carbon Society, a global issue”: Realization of a low carbon society through game changing technologies. Research title: ‘Creation of PEP-accumulating chassis strains by cell surface engineering and metabolic engineering’ (Grant No. JPMJMI17EI).
Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (B) (Grant No. 19H02526)
RIKEN Center for Sustainable Resource Science, Special Postdoctoral Researcher Program.
Grant-in-Aid for JSPS Research Fellows
Journal Information
Nature Communications
Title “Metabolic engineering of Escherichia coli for shikimate pathway derivative production from glucose–xylose co-substrate”
DOI:10.1038/s41467-019-14024-1
Authors: Ryosuke Fujiwara, Shuhei Noda, Tsutomu Tanaka, Akihiko Kondo
Source
Kobe University, press release, 2020-02-25.
Supplier
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals









