Carbon monoxide is an insidious poison because it loves the iron in our blood; it pushes oxygen out of iron-based hemoglobin, leading to painful asphyxiation.
This affinity for iron comes in handy in a newly created material that can absorb carbon monoxide far better than other materials, with potential applications in industrial processes like syngas production, where CO is a key player, and reactions where CO is an unwanted contaminant.
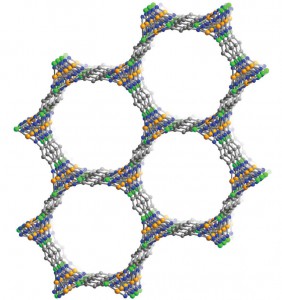
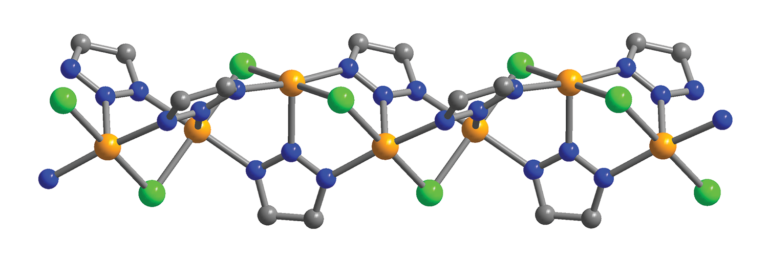
The new material is a metal-organic framework – an amazingly porous material with a growing list of applications – that incorporates chains of iron atoms tuned to attract CO and exclude other chemical compounds. When CO binds to an iron atom in the MOF, it changes the environment of neighboring iron atoms to make them even more attractive to CO, creating a chain reaction.
“We see this cooperative adsorption effect where binding at one site activates the neighboring sites, which means that all of a sudden you go from very little adsorption to essentially saturating the material with CO,” said senior researcher Jeffrey Long, a UC Berkeley professor of chemistry and faculty scientist at Lawrence Berkeley National Laboratory.
The CO binding flips the spin state of iron, hence Long’s terminology for the material: spin-transition MOFs.
Two years ago, Long accidentally stumbled across the first of this type of cooperative adsorbent when he created a MOF that adsorbed carbon dioxide far better than other materials.
“The carbon dioxide capture material we lucked into in 2015 was a first-of-its-kind material for cooperative adsorption,” he said. “Now we’ve shown that cooperative MOF adsorbents can be built by design to target other key industrially relevant molecules for separation. It is a fundamental new mechanism where, by adjusting the ligands bound to the iron, you might be able to get unsaturated hydrocarbons like acetylene, ethylene and propylene to bind also.”
The research, posted online Sept. 11 in advance of publication in the journal Nature, was supported by the Center for Gas Separations Relevant to Clean Energy Technologies, an Energy Frontier Research Center operated jointly by UC Berkeley and Berkeley Lab and funded by the U.S. Department of Energy.
Recovering rather than burning carbon monoxide
CO is used in a variety of industrial processes, including as a component of synthesis gas – a mix of CO and hydrogen used to make synthetic fuel or to synthesize other chemicals. These MOFs might serve as reservoirs for CO to maintain the correct ratio of CO to hydrogen for a particular reaction.
CO is also an essential intermediate in iron and steel production. Long predicts that the new MOF could be used to extract CO from the mixed-gas byproducts of such manufacturing to provide recycled CO for reuse in the chemical industry. In most cases today, these mixed gases are burned, Long said, accounting for a large portion of the greenhouse gases produced by the steel industry.
Such MOFs also could help suck up CO in reactions where CO poisons the catalyst, such as in the production of ammonia for fertilizers or polymers like polyethylene and polypropylene, and in hydrogen fuel cells.
“There are lots of places where you want to separate CO sufficiently in industry, and these spin transition MOFs can potentially have a role there,” Long said.
In practice, the MOFs would adsorb CO at room temperature, and then be heated slightly to drive off the CO, readying the MOF for reuse. These spin-transition MOFs can be precisely tuned so that only a small rise in temperature – from 20 C to 60 C, for example — releases the CO, requiring significantly less energy than other capture or storage technologies, such as cryogenic distillation.
As an example, they compared their spin-transition MOF to a commercial, liquid absorbent process for recovering CO, which is called COSORB. Initial calculations showed that the MOF requires just 32 percent of the energy to capture and reuse CO as the COSORB process.
Co-authors of the paper are Douglas Reed, Benjamin Keitz, Julia Oktawiec, Jarad Mason, Tomče Runčevski, Dianne Xiao and Lucy Darago of UC Berkeley and Valentina Crocellà and Silvia Bordiga of the University of Turin.
Author
Robert Sanders
Source
University of California, Berkeley, press release, 2017-09-11.
Supplier
Lawrence Berkeley National Laboratory
University of California, Berkeley (UC Berkeley)
US Department of Energy (DoE)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










