Not only do the practitioners have to break down the complex biomass mixtures efficiently to synthesize bio-ethanol, but they also have to contend with numerous other equally (and often times more) important considerations such as low oil prices (per barrel), process safety, waste streams, and chemical toxicity, recycling of solvents/catalysts, process economics, and a multitude of engineering/ technology considerations.
Nevertheless, despite the challenges, the ideal outcome of these efforts when accomplished is quite satisfying: a simple, efficient, green, robust, and safe manufacturing process. A showcase of green chemistry, process intensification, and catalysis along with industrial fermentation – Butanol production (with a global market of about 350 million gallons per year) has garnered wide acclaim.
Butanol is an important industrial chemical, which is currently produced by the Oxo-process starting from propylene with hydrogen and carbon monoxide (usually in the form of synthesis gas) over an expensive rhodium catalyst or the Aldol-process starting from acetaldehyde, this is usually referred to as the – petroleum-derived approach as the components i.e., propylene and synthesis gas are derived from petroleum/fossil sources and hence inherently “not green”. The Oxo Process was developed and licensed to the industry through a tripartite collaboration beginning in 1971. The principals were Johnson Matthey & Co. Ltd. (now Johnson Matthey PLC), The Power-Gas Corporation Ltd. (a former name of Davy Process Technology Ltd., now a subsidiary of Johnson Matthey PLC) and Union Carbide Corporation (now a subsidiary of The Dow Chemical Company).
The second, greener pathway is the ABE-fermentation process (Acetone, Butanol, Ethanol) that was pioneered by Chaim Weizmann during World War I. At the time, the petroleum-derived approach proved to be economically advantageous in comparison to the ABE-fermentation based processes. For this reason, most of the facilities using butanol / acetone fermentation process ceased to operate with a few exceptions in Mainland China and South Africa (until 1980s). This happened after World War II when rapid development of the petrochemical industry took place.
However, with crude oil prices fluctuations coupled with other geopolitical considerations across the globe have sparked an urgent interest in achieving a transition from non-renewable carbon resources to renewable bio-resources. In addition, researchers and start-ups from across the globe have reported on their staggering progress towards research and development efforts towards the production of butanol from renewable resources. The ABE-process has since received wide acceptance for a second come back to match mandates around the world to meet the standard blends with the petroleum counterparts. Some start-ups have also claimed their niche in developing butanol from the ubiquitous carbon dioxide, or from Scotch whisky by-products! Often the positive points are highlighted – sometimes exaggerated while the negative ones are not openly discussed such as the inherent toxicity of butanol to the cell wall of the microorganism synthesizing it in addition to the low yields.
Nonetheless, a plethora of start-ups and joint ventures flourished since early 2000 and the numbers are growing steadily. The number of start-ups pursuing various aspects of butanol is flourishing more than ever – from carbon dioxide capture, to developing alcohol-to-jet (AtJ) fuel with the potential to deliver aviation biofuels, developing new microbial strains for efficient conversions to butanol, feedstock analysis, to downstream processing. With this astounding pace one would not be surprised to find a start-up next door aiming to be the next analogue of the Oxo-process to synthesize 100% of global butanol production using renewable resources!
Butanol has since successfully made the transition from a commodity chemical to a fuel additive specially when it is compared to a much hygroscopic and less energy dense – ethanol. Butanol is versatile for many reasons – one could use it as a fuel additive or chemically transform it to high value high volume precursors such as butanal (global production around 6.6 x 106 tons/year), glycol ethers, butyl acrylates, solvents – butanol by itself can be used as a solvent or converted to the more widely used (“workhorse”) plasticizer such as 2-ethyl hexanol (global production around 2.5 x 106 tons/year) etc. So, contemplating a niche in downstream processing to focus on renewable chemicals or plastics, one may consider butanol conversion to lucrative chemicals markets.
One may choose to go to the other end of the spectrum and consider making alcohol-to-jet (AtJ) fuel blend with the ethanol or butanol platform. For example, a plane like a Boeing 747 consumes approximately 1 gallon of fuel (about 3.78 liters) per second. Over the course of a 10-hour flight, (say from Berlin, Germany to New York, United States) it burns about 36,000 gallons (150,000 liters). The Boeing 747 consumes approximately 5 gallons of fuel per mile (12 liters per kilometer). So you would make multitude volumes of the fuel to meet the blending requirements in the aviation department.
With the right tools in your belt this is certainly achievable – but remember the goal is to blend NOT replace petroleum 100%!
Exhibit 1.
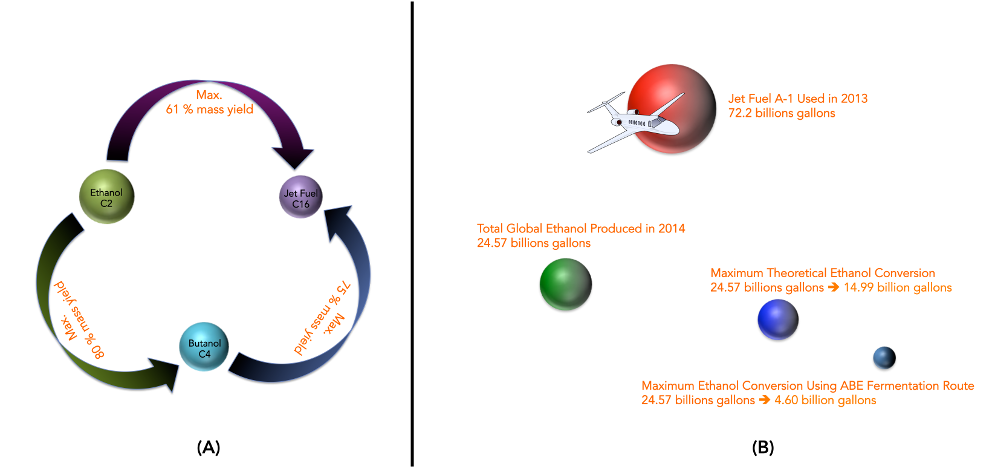
The annual consumption of Jet A-1 fuel used by commercial operators according to the Air Transport Action Group (ATAG) in 2013 was 72.2 billion gallons. The annual global production of ethanol according to the RFA Analysis of Public & Private Estimates was 24.57 billion gallons in 2014, Exhibit 1. In considering the de novo design of any systems that upgrade existing biofuels (such as ethanol to n-butanol or to Jet Fuel A-1) it is imperative to design working systems that are relatively inexpensive to keep investments low which is reflected in the pricing of the end product, i.e., a fuel, ethanol to an upgraded fuel, n-butanol or Jet Fuel A-1 as contemplated here. A simple back of the envelope calculation would suggest the following: with 100% conversion of ethanol to n-butanol the mass yield would be 80%. Similarly, for 100% conversion of ethanol to C16 hydrocarbon (Jet Fuel A-1, contains C8-C16 i.e., carbon atoms per molecule) the mass yield would be 61% and if you started from butanol, the mass yield would be 75% of Jet Fuel A-1.
So theoretically if ALL of ethanol produced globally were to be converted to Jet Fuel A-1 i.e., 24.57 billion gallons one will obtain 15 billion gallons of Jet Fuel A-1. Recall the need from year 2013 was 72.2 billion gallons! This amounts to only 21% of the requirement and therefore may be used only as a blend and NOT a replacement.
Where does the ABE fermentation stand – The best results ever obtained for the ABE fermentations to date are in the vicinity of 20 g/L in butanol concentration from fermentation, 4.5 g/L/h in butanol productivity, and a butanol yield of less than 25% (w/w) from glucose (1.29 gallons per bushel where, 1 US bushel = 35.2391 L). Therefore if the fermentation route were to be utilized – the limiting step would be a conversion of ethanol to butanol. Thus, 24.57 billion gallons of ethanol would give only 4.6 billion gallons of Jet Fuel A-1, which amounts to 6.4% of the actual requirement of the commercial operators.
If you are a smaller/medium sized start-up with a novel idea working on designing a proof-of-concept pilot before venturing into a full scale demo or commercial plant or just planning to expand your patent portfolio or a big conglomerate planning to be brand ambassadors for developing sustainable solutions for tomorrow based on the butanol platform you would want to consider a range of factors including market studies, and important considerations such as value/volume chain analysis, technology know how, process economics, industrial fermentation, separation technology, green chemistry, engineering/technology, or arranging off-take agreements with customers, local and offshore, design and fabrication of pilot plants; region specific legislations associated with the blending protocols, related documentation with RINs credits etc.
The list certainly seems monumental but given most start-ups in the field are about to set up a chain of events that will be disruptive in how we as a generation look at energy and its utility – It is definitely a challenge that needs a global perspective and it is most certainly not trivial!
Source
The Energy Collective, 2015-01-04.
Supplier
Air Transport Action Group (ATAG)
Dow Chemical Company
Johnson Matthey
Lee Enterprises Consulting
Renewable Fuels Association (RFA)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals










