Diesel and petrol cars as well as trucks, airplanes, and ships could run on such fuels in a climate neutral manner. They can also be used as energy storage systems, which could help compensate the fluctuations of wind and solar energy. As part of the PROMETHEUS project, Jülich researchers together with WZR ceramic solutions GmbH, the Greek Aristotle University of Thessaloniki, and the mineral oil group Hellenic Petroleum want to develop a 3D-printed membrane reactor with ultrathin cells for the production of synthetic fuels. This reactor is expected to be considerably more efficient and cost-effective than current devices, which, for the most part, are still at an experimental stage.

Copyright: Forschungszentrum Jülich / T. Schlößer
Synthetic fuels are a potential solution for powering combustion power plants and engines as well as industrial plants in a climate-friendly manner – if the necessary energy stems from renewable sources. When these are combusted, they only release the CO2 that was bound during their production. Synthetic fuels are by nature pure and emit virtually less pollutants during combustion. Nitrogen oxides and fine dust are thus not released in any significant amounts. Since they are also easy to transport and store, they are suitable for use as energy storage systems for the transformation of the German energy sector (Energiewende). If fed into the gas grid, they can be converted back into electricity in gas power plants as needed, for example whenever the sun and wind do not provide sufficient energy.
The prices of synthetic fuels are, however, still quite high and represent an obstacle to the production of large quantities. Co-electrolysis is a relatively new method and a promising option for reducing the production costs. It is viewed as a very efficient method, but is still in its infancy.

Copyright: Forschungszentrum Jülich / T. Schlößer
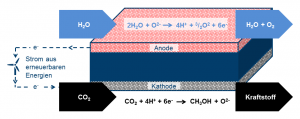
The new technology makes it possible to produce both synthetic chemicals and fuels directly in one step. Current methods, in contrast, require several process steps. Within the PROMETHEUS project, Jülich researchers want to develop a membrane reactor for co-electrolysis in which several chemical reactions are possible. The core element is a ceramic membrane that is permeable to hydrogen and oxygen ions. There are catalyst layers on its surfaces that speed up the process of the desired conversion reactions.
3D printing for tailor-made designs
“The efficiency of the process depends on several factors, including membrane thickness, surface activity, and the porosity of the substrate. These will be optimized in the project,” explains Prof. Wilhelm Meulenberg from Jülich’s Institute of Energy and Climate Research (IEK-1), who heads the project. To increase the flow through the membrane, the researchers have designed it as an ultrathin layer. At 10–50 micrometres, it is about as thin as a human hair. The thinner the material, the lower the transport resistance, and the more hydrogen can pass through the membrane at the same time.

Copyright: Forschungszentrum Jülich
To achieve the necessary mechanical stability, the membrane layer is deposited on a much thicker porous substrate. The use of 3D printing for ceramics, which is the area of expertise of WZR ceramic solutions GmbH, offers several advantages: “On the one hand, these processes permit the production of a substrate with a tailor-made pore structure with pore channels optimized for gas transport. On the other hand, 3D printing also contributes to significantly reducing the subsequent costs of producing membrane cells in comparison to multistage casting and sintering processes,” says Meulenberg.
WZR ceramic solutions GmbH develops a number of methods of additive manufacturing (3D printing) for industrial applications. “In addition to the geometric freedom of the new techniques, the option of modifying or combining materials on a microscopic level opens up the opportunity for completely new products. No other process enables the local modification and exact adaptation of a ceramic structure to the specific requirements of a component. We want to exploit this potential to produce optimized substrates,” adds Wolfgang Kollenberg, CEO of WZR ceramic solutions GmbH.
The project will receive € 350,000 in funding over three years from the Federal Ministry of Education and Research (BMBF) as part of the German–Greek Research and Innovation Programme. If all goes well, the researchers will present a design by the end of the project for a functional low-cost membrane reactor which can be used for synthesizing methane (CH4), methanol (CH3OH), and syngas (CO+H2). One potential end user of the technology is already involved: ELPE Renewables (ELPE RES) is the renewables section of the Greek mineral oil group Hellenic Petroleum (HELPE) and will conduct a feasibility and implementation study for the project.
How does a membrane reactor work?
Copyright: Forschungszentrum Jülich
Source
Forschungszentrum Jülich, press release, 2018-09-25.
Supplier
Aristoteles-Universität Thessaloniki
Forschungszentrum Jülich
WZR ceramic solutions GmbH
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals









